ARG22203
anti-Amyloid Oligomers (A11) antibody
anti-Amyloid Oligomers (A11) antibody for Dot blot,ELISA,ICC/IF,IHC-Formalin-fixed paraffin-embedded sections,Immunoprecipitation,Western blot and Human,Mouse,Rat
概述
| 产品描述 | Rabbit Polyclonal antibody recognizes Amyloid Oligomers (A11) |
|---|---|
| 反应物种 | Hu, Ms, Rat |
| 应用 | Dot, ELISA, ICC/IF, IHC-P, IP, WB |
| 特异性 | Recognizes all types of amyloid oligomers. Appears to recognize a peptide backbone epitope that is common to amyloid oligomers, but is not found in native proteins, amyloidogenic monomer. |
| 宿主 | Rabbit |
| 克隆 | Polyclonal |
| 靶点名称 | Amyloid Oligomers (A11) |
| 抗原物种 | Human |
| 抗原 | Synthetic molecular mimic of soluble oligomers |
| 偶联标记 | Un-conjugated |
| 別名 | AAA; AD1; PN2; ABPP; APPI; CVAP; ABETA; PN-II; CTFgamma |
应用说明
| 应用建议 |
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 应用说明 | * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
属性
| 形式 | Liquid |
|---|---|
| 纯化 | Purification with Protein A. |
| 缓冲液 | PBS, 0.09% Sodium azide and 50% Glycerol |
| 抗菌剂 | 0.09% Sodium azide |
| 稳定剂 | 50% Glycerol |
| 存放说明 | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| 注意事项 | For laboratory research only, not for drug, diagnostic or other use. |
生物信息
| 数据库连接 | |
|---|---|
| 基因名称 | APP |
| 全名 | amyloid beta precursor protein |
| 细胞定位 | Membrane |
| 预测分子量 | 87 kDa. (79 - 120 kDa depending on glycosylation level) |
| 翻译后修饰 | Proteolytically processed under normal cellular conditions. Cleavage either by alpha-secretase, beta-secretase or theta-secretase leads to generation and extracellular release of soluble APP peptides, S-APP-alpha and S-APP-beta, and the retention of corresponding membrane-anchored C-terminal fragments, C80, C83 and C99. Subsequent processing of C80 and C83 by gamma-secretase yields P3 peptides. This is the major secretory pathway and is non-amyloidogenic. Alternatively, presenilin/nicastrin-mediated gamma-secretase processing of C99 releases the amyloid beta proteins, amyloid-beta 40 (Abeta40) and amyloid-beta 42 (Abeta42), major components of amyloid plaques, and the cytotoxic C-terminal fragments, gamma-CTF(50), gamma-CTF(57) and gamma-CTF(59). Many other minor beta-amyloid peptides, beta-amyloid 1-X peptides, are found in cerebral spinal fluid (CSF) including the beta-amyloid X-15 peptides, produced from the cleavage by alpha-secretase and all terminating at Gln-686. Proteolytically cleaved by caspases during neuronal apoptosis. Cleavage at Asp-739 by either caspase-6, -8 or -9 results in the production of the neurotoxic C31 peptide and the increased production of beta-amyloid peptides. N- and O-glycosylated. O-glycosylation on Ser and Thr residues with core 1 or possibly core 8 glycans. Partial tyrosine glycosylation (Tyr-681) is found on some minor, short beta-amyloid peptides (beta-amyloid 1-15, 1-16, 1-17, 1-18, 1-19 and 1-20) but not found on beta-amyloid 38, beta-amyloid 40 nor on beta-amyloid 42. Modification on a tyrosine is unusual and is more prevelant in AD patients. Glycans had Neu5AcHex(Neu5Ac)HexNAc-O-Tyr, Neu5AcNeu5AcHex(Neu5Ac)HexNAc-O-Tyr and O-AcNeu5AcNeu5AcHex(Neu5Ac)HexNAc-O-Tyr structures, where O-Ac is O-acetylation of Neu5Ac. Neu5AcNeu5Ac is most likely Neu5Ac 2,8Neu5Ac linked. O-glycosylations in the vicinity of the cleavage sites may influence the proteolytic processing. Appicans are L-APP isoforms with O-linked chondroitin sulfate. Phosphorylation in the C-terminal on tyrosine, threonine and serine residues is neuron-specific. Phosphorylation can affect APP processing, neuronal differentiation and interaction with other proteins. Phosphorylated on Thr-743 in neuronal cells by Cdc5 kinase and Mapk10, in dividing cells by Cdc2 kinase in a cell-cycle dependent manner with maximal levels at the G2/M phase and, in vitro, by GSK-3-beta. The Thr-743 phosphorylated form causes a conformational change which reduces binding of Fe65 family members. Phosphorylation on Tyr-757 is required for SHC binding. Phosphorylated in the extracellular domain by casein kinases on both soluble and membrane-bound APP. This phosphorylation is inhibited by heparin. Extracellular binding and reduction of copper, results in a corresponding oxidation of Cys-144 and Cys-158, and the formation of a disulfide bond. In vitro, the APP-Cu(+) complex in the presence of hydrogen peroxide results in an increased production of beta-amyloid-containing peptides. Trophic-factor deprivation triggers the cleavage of surface APP by beta-secretase to release sAPP-beta which is further cleaved to release an N-terminal fragment of APP (N-APP). Beta-amyloid peptides are degraded by IDE. |
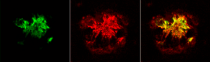
检测图片 (1) Click the Picture to Zoom In
-
ARG22203 anti-Amyloid Oligomers (A11) antibody IHC image
Immunohistochemistry: Formalin-fixed Human Alzheimer's Disease brain stained with ARG22203 anti-Amyloid Oligomers (A11) antibody at 1:1000. Secondary Antibody: Goat anti-Rabbit ATTO 594 (red). (A) ARG22204 anti-Amyloid Fibrils (OC) antibody. (B) ARG22203 anti-Amyloid Oligomers (A11) antibody. (C) Composite.